Translate this page into:
Post-operative Hemorrhage After Tumor Removal of Multiple Meningiomas
* Corresponding author: Dr. Bob Irfan Syahputra, Department of Neurosurgery, Padjajaran University, Bandung, Indonesia. bobirfansyahputra@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Syahputra BI, Imron A, Febriantara D, Efriani H. Post-operative Hemorrhage after Tumor Removal of Multiple Meningiomas Int J Recent Surg Med Sci. 2025;11(e005). doi: 10.25259/IJRSMS_4_2025
Abstract
Post-resection hemorrhage is a rare but serious complication of meningioma surgery. In this case report, we present a rare case of a patient with multiple meningiomas who developed intracranial hemorrhage (ICH) following tumor resection. In this case, A 51-year-old woman with multiple meningiomas presented to our department with severe recurrent headaches. The patient underwent bilateral craniotomy and cranioplasty, successfully reducing a total of 80% of the meningioma mass. However, two days post-surgery, her mental status deteriorated, and a CT scan revealed spontaneous intracranial hemorrhage (ICH); the patient cito operated for the evacuation with craniotomy. The patient showed improvement and was sent home after seven days of medical treatment in the hospital. Though rare, intracranial hemorrhage following meningioma resection is a life-threatening complication. Case reports or series can provide valuable insights for clinicians when managing such complications.
Keywords
Intracranial hemorrhage
Meningioma
Multiple meningiomas
Tumor-related hemorrhage
INTRODUCTION
Meningioma is one of the most common primary brain tumors, representing approximately 30% of all primary brain tumors. Meningiomas arise from meningothelial cells (MECs) in the brain membranes, primarily from the cellular components of the pia mater, arachnoid mater, and trabeculae and septa of the subarachnoid space.[1-3] Histologically, MECs are the cell layers responsible for the physical barrier of the central nervous system and also play a significant role in triggering or dampening immune responses in the central nervous system through the secretion of pro- or anti-inflammatory cytokines.[2-8]
Meningiomas are usually slow-growing tumors, most of which are benign and asymptomatic, but in rare cases, they can become malignant. The etiology of meningiomas is unknown, but some potential causative factors include exposure to ionizing radiation, obesity, occupational herbicide exposure, and hormone therapy.[9-13] The most common risk factor is radiation exposure, especially to therapeutic radiation. Individuals who have received therapeutic radiation for other conditions, such as head and neck cancer, are at increased risk for developing meningiomas. Neurofibromatosis type 2 (NF2) is a genetic disorder associated with a higher risk of meningioma. A family history of meningiomas is also a risk factor for developing the tumor.[5,9,14,15]
Meningiomas arise from the arachnoid cells lining the dura mater. These cells proliferate and form a mass that presses on the surrounding brain tissue. Meningiomas are classified into three grades based on their histological characteristics.[5,14,16-19] Grade I meningiomas are benign tumors, while grade II and III meningiomas are malignant tumors. Grade I meningiomas account for about 90% of all meningiomas and have a low recurrence rate. Grade II and III meningiomas account for about 10% of all meningiomas and have a higher recurrence and mortality rate.[5,8,15] Symptoms of meningioma depend on the location and size of the tumor. Meningiomas can cause headaches, seizures, weakness or numbness in the hands or feet, vision problems, hearing problems, and cognitive changes. Symptoms of a meningioma can be nonspecific and mimic other neurological conditions, complicating diagnosis. In some cases, meningiomas may be asymptomatic and are discovered incidentally on imaging tests.[2,7,10,14,17,20]
Multiple meningioma
Multiple meningiomas are conditions in which there are >2 spatially separated synchronous or metachronous lesions in a single patient. This condition occurs in only 1%-10% of cases. Multiple meningiomas can be sporadic, radiation-induced, familial [associated with Neurofibromatosis Type 2 (NF2)], or familial meningiomatosis.[1-3,14]
Multiple meningiomas are more difficult to manage because surgical removal of multiple tumors may not be possible in a single incision. This can increase the complication rate. Surgical removal is the primary treatment for multiple meningiomas because half of patients with multiple meningiomas are symptomatic at presentation due to the tumor mass effect.[2,9,10,14]
CASE REPORT
A 51-year-old woman with blurred vision for 2.5 years, accompanied by behavioral disorders, was brought to the hospital. She underwent a contrast head CT scan, which showed multiple masses on the bilateral and right frontal bases [Figure 1 and 2].
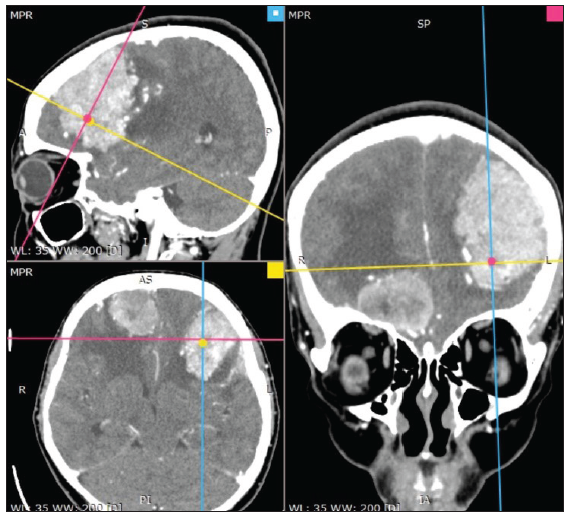
- Pre-op Contrast Head CT Scan showing 3 masses of the frontal: The first in the right frontal skull base, the second located in the right frontal posteriorly, third located in the left frontal. All the masses show enhanced homogeneously with contrast administration and dural tail. The red line a cut or slice that showing coronal View (right side). The blue line a cut or slice that showing sagital view (top left). The yellow line a cut or slice that showing axial View (bottom left). Dot showing marker position squares blue (sagital), red (coronal), and yellow (axial).
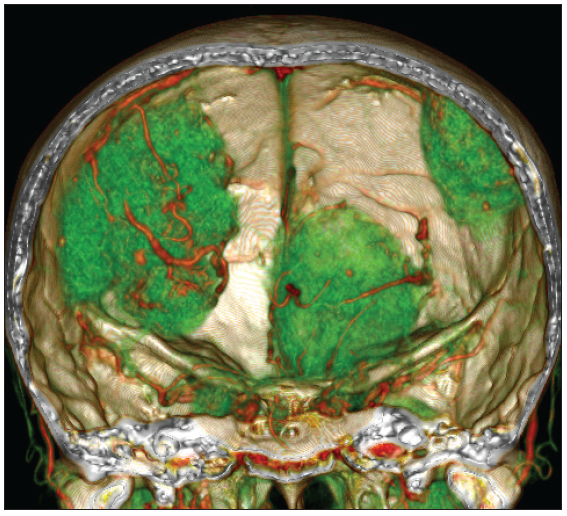
- 3D CT Reconstruction: Tumor appears reconstructed in green, showing vascularization surrounding the tumor. The red color in the 3D reconstruction shows vascularization as a tumor feeder.
In the angiography Study of the head CT angio reconstruction, there was a feeder artery in the left frontal mass, originating from the pial with the M4 segment running transversely on the lateral part of the tumor, and the trolar vein posterior to the tumor. In the right frontal mass, there was a feeder artery originating from the pial with the position of the trolar vein being superior to the tumor. In the right frontal base mass, there was a feeder artery from the anterior ethmoidal and branches from A2 and A3 [Figures 3 and 4].
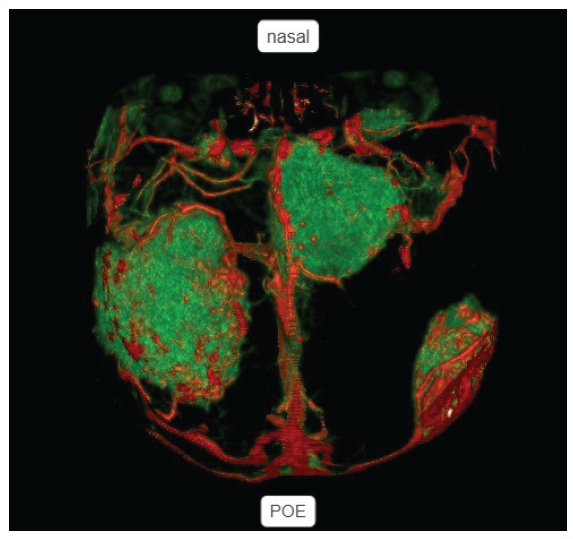
- 3DCT Reconstruction in the reversed position: On the left side showing nasal side located superiorly, superior sagittal sinus (SSS) located posteriorly, and protuberence occipital externa (POE) located inferiorly. Green color shows tumor mass on bilateral frontal, and red color shows vascularization.
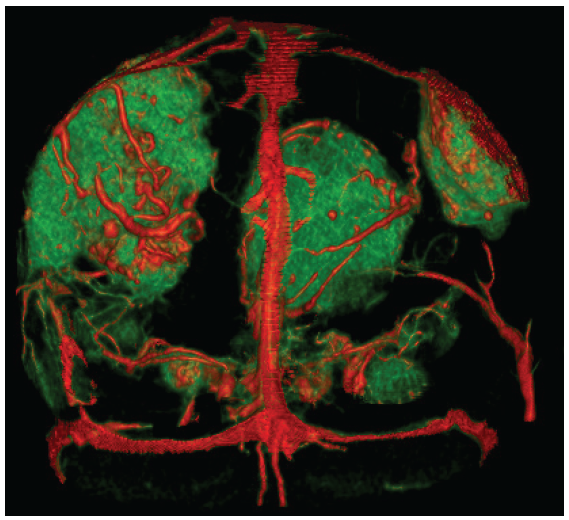
- 3DCT Reconstruction showing from posteriorly to anteriorly. Green color shows tumor mass on bilateral frontal, and red color shows vascularization.
The patient underwent tumor removal surgery with a craniotomy (CTR). A bifrontal incision was chosen because the tumor was located in both the frontal areas (green tumor mass projection markers visible) [Figure 5]. We decided on a 1-stage tumor removal surgery considering that the three tumors were easily accessible, superficial, and estimated to be safe enough from important vascularization structures, and the meningioma substance was considered.
![Positioning, Tumor marking, and Design of Incision: The patient with supine in the neutral head position, both pictures show a green marker (tumor), bicoronal incision (black line), estimated coronal suture (black dot line), and midline [black dot line from nasion to protuberence occipital externa (POE)].](/content/157/2025/11/1/img/IJRSMS-11-e005-g5.png)
- Positioning, Tumor marking, and Design of Incision: The patient with supine in the neutral head position, both pictures show a green marker (tumor), bicoronal incision (black line), estimated coronal suture (black dot line), and midline [black dot line from nasion to protuberence occipital externa (POE)].
The tumor mass was found to be white-grey, difficult to suction, with easy bleeding. The tumor in the left and right frontal areas was removed with a combined removal of 70%, and 80% was left over the attached vascular structure and on the right frontal base [Figure 6].
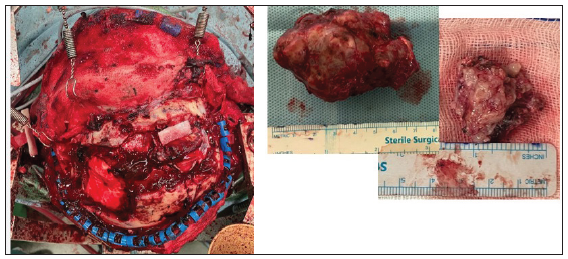
- Intraoperative finding: Left side showing after tumor removal, right side showing tumor with grayish white, rubbery, and difficult to suction.
However, on the 2nd postoperative day, the patient worsened with a change in mental status. Post-op CT scan evaluation showed a hyperdense lesion in the right frontal area, the initial possibility that we thought was hemorrhage from previously resolved venous seepage that drained into the trolar vein and another possibility from residual tumor hemorrhage. To rule out extracranial, we re-evaluated the blood test and blood clotting factors, but the result was within normal limits [Figure 7].
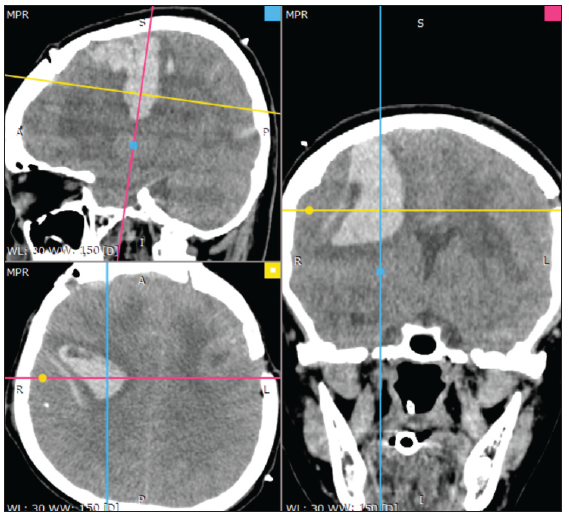
- Post-op CT Scan: Showing compression of the brain with hyperdense lesions in the right frontal, approximately 50cc with midline shift >10 mm. The red line a cut or slice that showing coronal view (right side). The blue line a cut or slice that showing sagital view (top left). The yellow line a cut or slice that showing axial view (bottom left). Dot showing marker position squares blue (sagital), red (coronal), and yellow (axial).
Then, the patient underwent craniotomy evacuation; we found a blood clot of about 40 cc with lysis of 5 cc with the source of bleeding from the capillaries of the tumor feeder after removal, which was seen during the operation, still experiencing seepage, evacuation, and hemostasis performed; the brain relaxed and pulsated. Primary Duroplasty was performed, and the skull was replaced with a titanium mesh [Figure 8].
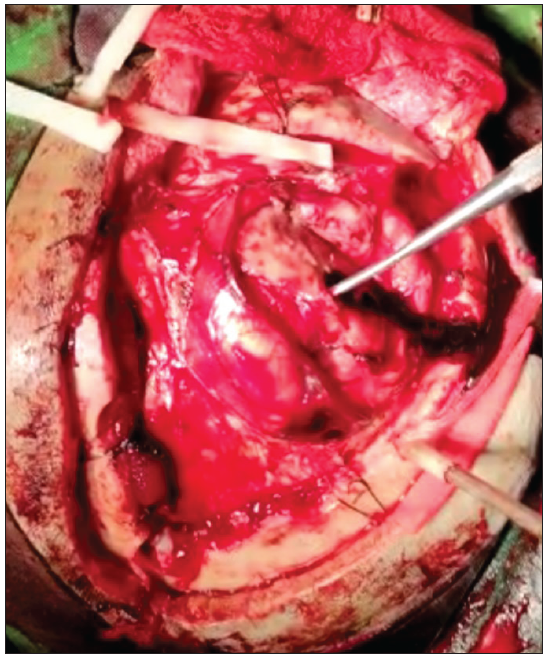
- Intraoperative intracerebral hemorhage (ICH) finding: After corticotomy showing brain relaxed and pulsating.
Post-operative (POD 2) evacuation, sedation, and glasgow coma scale (GCS) 15 (POD 3) were gradually ceased. The patient observed intracranial pressure until POD 7, and was sent home without new neurological deficits and visual analog scale (VAS) improvement of 1/10.
DISCUSSION
Postoperative bleeding is a significant complication of meningioma surgery. The incidence of postoperative bleeding after meningioma removal varies across studies, ranging from 2% to 23%. Disparities in incidence rates can be attributed to differences in surgical technique, tumor size and location, patient comorbidities, and other factors.[10,14,16,20]
Several factors increase the risk of bleeding after meningioma removal. These factors include tumor size, location, vascularity, and proximity to important structures.[2,3,5,16] Large tumors with extensive blood supply are more likely to bleed during and after the surgery. Tumors located in the parasagittal and skull base areas are also at higher risk of bleeding due to the complexity of the surgery and proximity to important structures such as blood vessels and nerves. In addition, patient factors such as hypertension, coagulation disorders, and use of anticoagulant medications can increase the risk of bleeding.[7,13,17]
Management of post-meningioma-removal bleeding depends on the severity of the bleed and the location of the hematoma. Mild bleeding can be managed conservatively with close monitoring of the patient’s vital signs and neurologic status.[7,17,20] Severe bleeding may require surgical intervention, including hematoma evacuation and control of the source of bleeding.[8,20]
In this patient, a craniotomy evacuation was performed for the identified bleeding area. She also underwent bipolar hemostasis accompanied with the administration of hemostatic agents. For the head CT scan and intraoperative findings, several operator limitations, like the location of the intracerebral hemorhage (ICH) on the side of the transverse trolar vein and in front of the central sulcus, were encountered. So, the operator could only maneuver to the angle from the inferior side of the corticotomy, because it is very susceptible to injury. Related to the cause of bleeding, several other theories also mention that changes in the vascularization structure of more than 60% in multiple meningiomas have been clearly proven, where the intima is more fragile in multiple meningiomas than in single-lesion meningiomas.[11,13,18,21] This is an important risk factor related to preoperative and intraoperative plans.
CONCLUSION
Bleeding after meningioma removal is a significant complication and can result in high morbidity and mortality. The incidence of bleeding varies depending on several factors, including the size, location, amount of mass, and vascularity of the tumor. Management of bleeding requires a multidisciplinary approach involving careful preoperative planning, intraoperative monitoring, and postoperative management. Preventing bleeding after meningioma removal is essential to reduce morbidity and mortality and requires careful attention to patient and tumor characteristics.
The best management neurosurgeon, besides the knowledge of the neurovascular anatomy involved from the tumor and the blood vessels around it, also ensures that all bleeding is handled well and carefully, which is the most crucial interest. Almost more or less more than 50% of every remaining tumor, even in a very small amount, is very likely to have the potential for bleeding. Some important measures taken after controlled bleeding are water irrigation and drainage until it looks like a very clear liquid. This is enough to ensure safety without bleeding.
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Population-based Analysis of Sporadic and Type 2 Neurofibromatosis-associated Meningiomas and Schwannomas. Neurology. 2000;54:71-6.
- [CrossRef] [PubMed] [Google Scholar]
- Genomic Sequencing of Meningiomas Identifies Oncogenic SMO and AKT1 Mutations. Nat Genet. 2013;45:285-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Multiple Meningiomas: A Clinical, Surgical, and Cytogenetic Analysis. Surg Neurol. 1989;31:255-60.
- [CrossRef] [PubMed] [Google Scholar]
- Multiple Meningiomas: Investigating the Molecular Basis of Sporadic and Familial Forms. Int J Cancer. 2003;103:483-8.
- [CrossRef] [PubMed] [Google Scholar]
- Clearance of Neurotoxic Peptides and Proteins by Meningothelial Cells. Exp Cell Res. 2020;396:112322.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical and Histological Features of Multiple Meningiomas Compared with Solitary Meningiomas. Neurol Res. 2005;27:324-32.
- [CrossRef] [PubMed] [Google Scholar]
- Meningioma: A Review of Clinicopathological and Molecular Aspects. Front Oncol. 2020;10:579599.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Clinicopathologic Features of Incidental Meningiomas: A Review of the Literature and the university of Rochester Autopsy Experience. Clin Neuropathol. 2019;38:118-21.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of a Progenitor Cell of Origin Capable of Generating Diverse Meningioma Histological Subtypes. Oncogene. 2011;30:2333-44.
- [CrossRef] [PubMed] [Google Scholar]
- Molecular Characteristics of Meningiomas. J Pathol Transl Med. 2020;54:45-63.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Meningothelial Cells as part of the Central Nervous System Host Defence. Biol Cell. 2013;105:304-15.
- [CrossRef] [PubMed] [Google Scholar]
- The 2007 WHO Classification of Tumors of the Central Nervous System. Acta Neuropathologica.. 2007;114:97-109.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Incidental Meningiomas in Autopsy Study. Surg Neurol. 1987;27:319-22.
- [CrossRef] [PubMed] [Google Scholar]
- CBTRUS Statistical report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Neuro Oncol. 2019;21:v1-v100.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Molecular Pathogenesis of Meningiomas. J Neurooncol. 2004;70:183-202.
- [CrossRef] [PubMed] [Google Scholar]
- Paediatric Meningiomas in Singapore — Case Series of a Rare Entity. J Clin Neurosci. 2020;73:62-6.
- [CrossRef] [PubMed] [Google Scholar]
- Microscopic Morphology and Histology of the Human Meninges. Morphologie. 2005;89:22-34.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and Etiology of Meningioma. J Neurooncol. 2010;99:307-14.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]








