Translate this page into:
Effect of Two Different Temperature Settings on Patient Comfort Level during Respiratory Therapy by High-Flow Nasal Cannula
Address for correspondence Megha Soni, MBBS, DA, DNB Anaesthesia, Assistant Professor, Department of Anaesthesia, Operation Theater Complex 3rd floor, ESIC Medical College, NH-3, Faridabad, India (e-mail: meghasoniarora@gmail.com).
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background
Hypoxemic respiratory failure may require high flow O2 therapy (>15 L/m), which can be delivered using a high-flow nasal cannula (HFNC) device. There are three variables: FiO2, flow, and temperature that can be controlled while using HFNC. This study was planned to assess the degree of comfort level of patients at two different temperature settings.
Methods
A prospective interventional cross-over study was performed on 40 hypoxemic respiratory failure patients, who were on HFNC. Primary outcome was to assess patient comfort during HFNC therapy at two different temperatures (31 and 37°C), at the flow of 60 L/m for 30 minutes at each temperature setting, leaving FiO2 unchanged. After 30 minutes of each temperature setting, the comfort level was assessed using 11 score visual numerical scale (ranging from 0 to 10) together with other vital parameters.
Result
Patients were found to be more comfortable at temperature settings of 31°C than at 37°C (p-value < 0.05%). Also, an increase in blood pressure was observed at 37°C after completion of 30 minutes, which was statistically significant but not clinically significant.
Conclusion
Starting HFNC therapy at lower temperature gives better patient acceptance and reduces chances of failure due to discomfort.
Keywords
high-flow nasal canula
temperature
comfort level
Introduction
Oxygen (O2) therapy is the first step to prevent and manage hypoxemic respiratory failure. High flows (>15 L/m),1 such as 30 L/m and 60 L/m can be easily delivered using HFNC. It has shown to have better tolerance and comfort level than conventional devices.2–4
Different parameters of HFNC device (FiO2, flow, and temperature) can be controlled independently. There are studies comparing patient comfort level between NIV and HFNC but there is a need to know the optimal settings of HFNC to achieve adequate patient comfort level. So, we planned this study to compare the degree of patient comfort level at different temperature settings of HFNC.
Materials and Methods
This prospective interventional cross-over study was carried out after patient's informed written consent, Institutional Ethics Committee approval, and CTRI registration (CTRI number- CTRI/2021/07/035224). Confidentiality of subjects was maintained. The procedures followed were in accordance with the ethical standards of the institutional committee on human experimentation and with the Helsinki declaration of 1975, as revised in 2000.
A total of 40 non-sedated patients on HFNC with appropriate sized nasal cannula at a flow rate of 60 L/min (as per the inclusion and exclusion criteria) participated in the study.
Inclusion criteria – Patients admitted in the ICU with age between 18 and 65 years on HFNC (Optiflow, Fisher & Paykel Healthcare, Auckland, New Zealand) Exclusion criteria – Patients admitted in the ICU with age < 18 years and > 65 years, hemodynamic instability, altered mental status, patients on non-invasive ventilation, intubated or tracheostomized, pregnant patients, and febrile or hypothermic patients
The primary objective of the study was to assess all 40 patients for the degree of comfort using 11 score visual numerical scale (VNS) ranging from 0 to 10 at the end of 30 minutes, each for two different temperature settings.
Secondary objective was to compare the changes noted in the vital parameters such as heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), respiratory rate (RR), and SpO2, at the end of each step.
Group A – temperature setting of 31°C and flow at 60 L/min.
Group B – temperature setting of 37°C and flow at 60 L/min.
Patients were placed in a semi-recumbent position in a calm environment. Baseline parameters of all non-sedated patients already on HFNC (Fisher & Paykel Healthcare) who were on a flow of 60 L/m (as advised by the ICU physician) were recorded such as HR, SBP, DBP, RR and SpO2. Every patient included in the study, on HFNC, temperature was set to 31°C. Patients were allowed to stay calm, by their own self for duration of 30 minutes. After 30 minutes, vital parameters were recorded and VNS score was asked. Temperature settings were changed to 37°C. Again, patients were left undisturbed for a period of 30 minutes, following which, comfort level and vital parameters were assessed. All data were recorded by the personnel other than the person who set the temperature for the HFNC device.
Results
There were no drop outs in the study; comfort level of all recruited 40 patients could be assessed as proposed in the study. The data collected are expressed as mean ± standard deviation (SD) with 95% confidence level. Mean differences were calculated from readings at 31°C minus 37°C. Paired t-test was applied to compare the variables. A p-value < 0.05 was considered statistically significant.
As this was a cross-over study, patients allotted in each of the two groups were same; hence, mean age and sex were same for the two groups. The mean age was 44.6 ± 2.33 years, and out of the 40 subjects, 24 were males and 16 females.
Comfort level – Patients were found to be more comfortable in group A in comparison to those in group B with a mean difference of 2.9 VNS at two temperatures (p < 0.05%), which was clinically as well as statistically significant (►Table 1, ►Fig. 1).

- Visual numeric scale at 31°C and 37°C.
| Variable | Mean ± SD | Mean difference | p-Value |
|---|---|---|---|
| VNS (31°C) VNS (37°C) | 8.3 ± 0.60 5.4 ± 0.54 | 2.9 | 0.000 |
| HR (31°C) HR (37°C) | 84.2 ± 9.88 84.5 ± 9.03 | −0.3 | 0.634 |
| SBP (31°C) SBP (37°C) | 118.5 ± 9.57 120.5 ± 9.44 | −2 | 0.000 |
| DBP (31°C) DBP (37°C) | 76.7 ± 8.72 78.3 ± 7.63 | −1.5 | 0.005 |
| MAP (31°C) MAP (37°C) | 89.9 ± 8.24 91.8 ± 7.92 | −1.8 | 0.000 |
| SpO2 (31°C) SpO2 (37°C) | 97.8 ± 0.81 97.8 ± 0.66 | −0.025 | 0.830 |
| RR (31°C) RR (37°C) | 20.0 ± 1.42 19.9 ± 1.19 | 0.175 | 0.484 |
*Abbreviations: DBP, diastolic blood pressure; HR, heart rate; MAP, mean arterial blood pressure; RR, respiratory rate; SpO2, oxygen saturation; SBP, systolic blood pressure; SD, standard deviation; VNS, visual numeric scale.
†Temperature at which reading is noted in brackets.
‡Mean differences were calculated from readings at temperature of 31°C minus 37°C.
Vital signs – Group B showed an increase in SBP, DBP, and MAP after completion of 30 minutes as compared to group A (p < 0.05), which was statistically significant although mean differences were not that profound, showing no clinical significance (►Table 1, ►Figs. 2–4).
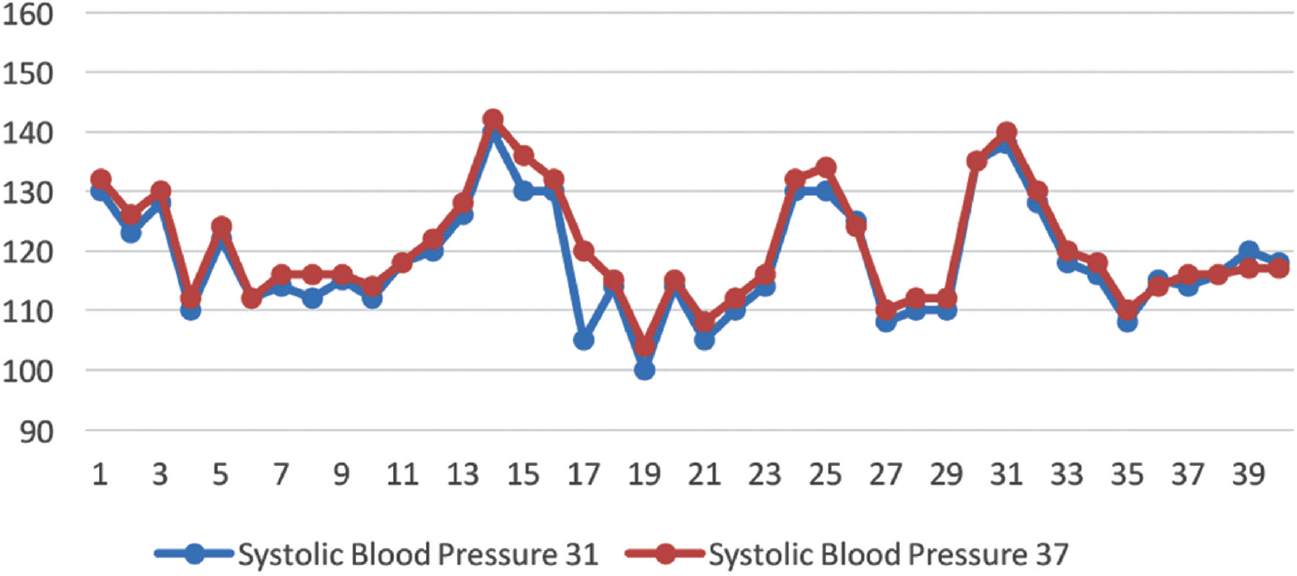
- Systolic blood pressure at 31°C and 37°C.

- Diastolic blood pressure at 31°C and 37°C.

- Mean arterial pressure at 31°C and 37°C.
Other variables such as HR, SpO2, and RR were not clinically as well as statistically significant (p > 0.05) (►Table 1, ►Figs. 5–7).

- Heart rate at 31°C and 37°C.

- Oxygen saturation at 31°C and 37°C.

- Respiratory rate at 31°C and 37°C.
Discussion
Earlier O2 therapy in spontaneously breathing patients were possible by nasal prongs and facemasks, which work well at flows of up to 10 L/m. High-flow O2 therapy (>15 L/m) may lead to drying of nasal mucosa even with bubble humidification due to air entrainment. Higher flows of totally conditioned gas (37°C containing 44 mg H2O/l (100% relative humidity)1 such as 30 L/m and 60 L/m can be delivered using HFNC.1 It prevents drying of mucosa, having a protective effect on muco-ciliary function such as secretion clearance and airway defense.
Also, gas conditioning may decrease inflammation of the tracheal mucosa after intubation; hence, the use of HFNC prior to extubation, prevents the administration of dry and cold air in the native airway of the patients, causing fewer reintubations secondary to upper airway obstruction and also, accelerated weaning in tracheostomized persons.2,5
Many studies have shown oxygenation enhancement and reduction in the respiratory rate by early application of HFNC compared to standard oxygen therapy.2–4 It has shown positive clinical impact on patients with acute hypoxemic respiratory failure.6–8 HFNC has better tolerance and comfort than non-invasive ventilation (NIV).9,10 HFNC reduces intubation rate, increasing ventilator-free days, and reduced 90-day mortality. However, the rate of intubation in patients on HFNC and NIV were found to be similar in many studies11–13 with studies showing better oxygenation with NIV.9,10
Better comfort levels help in improving the tolerance for any undergoing procedure. Recently, FLORALI study (post hoc analysis) showed intubation within 1 hour of starting HFNC due to discomfort, suggesting a strong link between discomfort and poor outcomes.14 Comfort level is a patient-level outcome generated on the basis of various physiologic mechanisms.15,16 Various physiological studies have been conducted before to measure comfort level in patients on non-invasive respiratory support, scale used was the same as in this study, i.e., VNS.17–19
In this study, patients were found to be more comfortable at lower temperature of 31°C than 37°C at 60 L/min of flow of HFNC. Marginal rise in SBP, DBP, and MAP was observed at higher temperatures, which was statistically significant but not clinically significant.
Comfort level is higher at lower temperatures and may suggest negative psychosomatic and physiologic signals such as excessive heating of nostrils in spite of the advantage of maximum humidity.17 However, because the heating and moisturizing function of the upper airway is preserved during use of the HFNC, starting HFNC at lower temperatures and gradually increasing it with time may be a considered, it may lead to better clinical outcomes such as better tolerance, longer duration of HFNC application, and improved comfort. Patients showed different comfort levels at different temperatures, may point toward personalizing the settings on individual basis rather than using fixed standard settings.20
Certain limitations pertinent to study are: firstly, our primary aim was subjective psychological outcome. Comfort level according to the temperature may be variable, and differ on person to person basis. Secondly, observation time in our study is limited to 30 minutes. Patient may show discomfort in a longer time of surveillance. Thirdly, ventilator settings including FiO2 were not considered in baseline comparisons, which may affect comfort levels. Fourthly, midway settings such as 34°C were not assessed, only extreme values were considered. Fifthly, larger studies with bigger sample size may validate our findings.
Conclusion
Starting HFNC therapy at lower temperature gives better patient acceptance and reduces chances of failure due to discomfort. Gradually temperature may be increased or adjusted on personalized basis according to the patients comfort. To use comfort level as a guide to HFNC settings as a non-invasive respiratory support, larger studies are needed for its validation.
Authors' Contributions
Dr. Mamta Kumari – concept, design, literature search.
Dr. Megha Soni- data acquisition, literature search.
Dr. Niharika Grover-literature search, definition of intellectual content, manuscript preparation.
Dr. Rashmi Taneja – manuscript editing and review.
Dr. Mamta Kumari is the guarantor and takes the responsibility for the integrity of work as a whole from inception to published article.
Declaration
This manuscript has been read and approved by all the authors, requirements for authorship have been met, and each author believes that the manuscript presents the honest work. Article is not published nor under consideration, in part or whole simultaneously in any other journal or any other proceedings.
Conflicts of Interest
None declared.
Funding
This study was funded by the ESIC Medical College, NH-3, Faridabad.
References
- High-flow nasal cannula support therapy: new insights and improving performance. Crit Care. 2017;21(01):62.
- [CrossRef] [PubMed] [Google Scholar]
- High-flow oxygen therapy in acute respiratory failure. Respir Care. 2010;55(04):408-413.
- [Google Scholar]
- Impact of high-flow nasal cannula oxygen therapy on intensive care unit patients with acute respiratory failure: a prospective observational study. J Crit Care. 2012;27(03):324.e9-324.e13.
- [CrossRef] [PubMed] [Google Scholar]
- Beneficial effects of humidified high flow nasal oxygen in critical care patients: a prospective pilot study. Intensive Care Med. 2011;37(11):1780-1786.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of postextubation highflownasal cannula vs conventional oxygen therapy on reintubation in lowrisk patients. A randomized clinical trial. JAMA. 2016;315(13):1354-1361.
- [CrossRef] [PubMed] [Google Scholar]
- High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015;372(23):2185-2196.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of high-flow nasal cannula on the work of breathing in patients recovering from acute respiratory failure. Crit Care Med. 2017;45(12):1981-1988.
- [CrossRef] [PubMed] [Google Scholar]
- Acute hypoxemic respiratory failure in immunocompromised patients: the Efraim multinational prospective cohort study. Intensive Care Med. 2017;43(12):1808-1819.
- [CrossRef] [PubMed] [Google Scholar]
- Sequential application of oxygen therapy via high-flow nasal cannula and noninvasive ventilation in acute respiratory failure: an observational pilot study. Respir Care. 2015;60(02):170-178.
- [CrossRef] [PubMed] [Google Scholar]
- Nasal high-flow oxygen therapy in patients with hypoxic respiratory failure: effect on functional and subjective respiratory parameters compared to conventional oxygen therapy and non-invasive ventilation (NIV) BMC Anesthesiol. 2014;14:66-73.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435-2441.
- [CrossRef] [PubMed] [Google Scholar]
- Noninvasive ventilation with helmet versus control strategy in patients with acute respiratory failure: a systematic review and meta-analysis of controlled studies. Crit Care. 2016;20:265-279.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of postextubation high-flow nasal cannula vs noninvasive ventilation on reintubation and postextubation respiratory failure in high-risk patients: a randomized clinical trial. JAMA. 2016;316(15):1565-1574.
- [CrossRef] [PubMed] [Google Scholar]
- REVA network. Predictors of intubation in patients with acute hypoxemic respiratory failure treated with a noninvasive oxygenation strategy. Crit Care Med. 2018;46(02):208-215.
- [CrossRef] [PubMed] [Google Scholar]
- Comfort Theory: a unifying framework to enhance the practice environment. J Nurs Adm. 2006;36(11):538-544.
- [CrossRef] [PubMed] [Google Scholar]
- A practical application of Katharine Kolcaba's comfort theory to cardiac patients. Appl Nurs Res. 2014;27(02):147-150.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of flow and temperature on patient comfort during respiratory support by high-flow nasal cannula. Crit Care. 2018;22(01):120.
- [CrossRef] [PubMed] [Google Scholar]
- BiPOP Study Group. High-flow nasal cannula therapy versus intermittent noninvasive ventilation in obese subjects after cardiothoracic surgery. Respir Care. 2017;62(09):1193-1202.
- [CrossRef] [PubMed] [Google Scholar]
- New setting of neurally adjusted ventilatory assist for noninvasive ventilation by facial mask: a physiologic study. Crit Care. 2017;21(01):170.
- [CrossRef] [PubMed] [Google Scholar]
- Systematic assessment of advanced respiratory physiology: precision medicine entering real-life ICU? Crit Care. 2017;21(01):143.
- [CrossRef] [PubMed] [Google Scholar]







